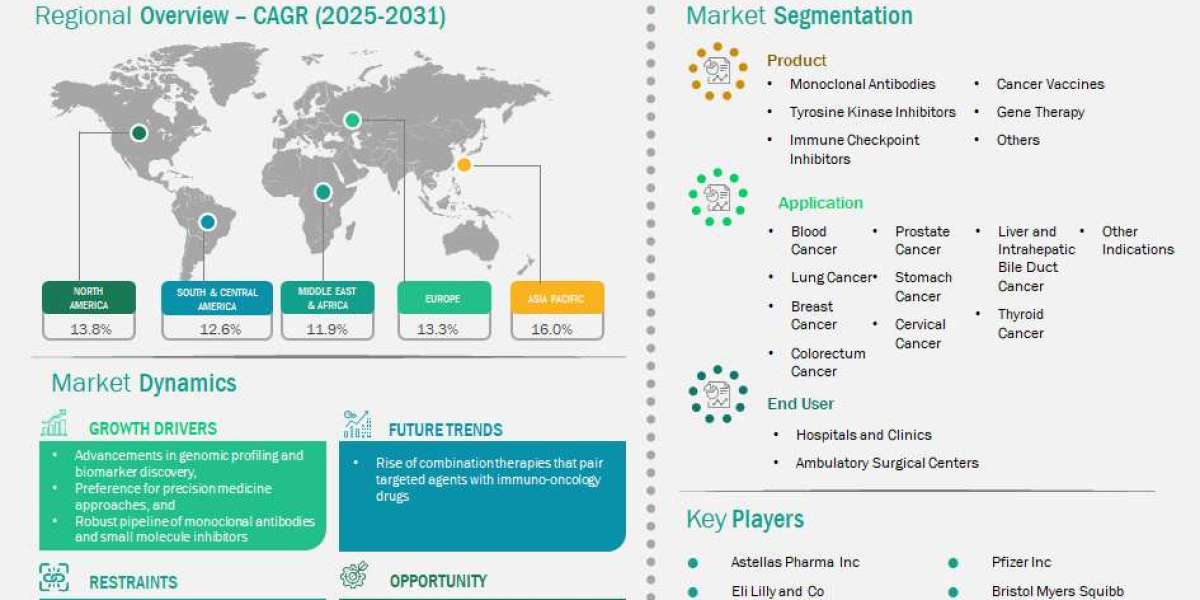
The Cancer Targeted Therapy Market presents one of the strongest revenue growth trajectories in the global pharmaceutical industry, and the Cancer Targeted Therapy Market Size figures reflect this exceptional expansion, underpinned by the irreversible global cancer burden with approximately 20 million new cases annually creating permanent structural therapy demand, AbbVie's May 2025 FDA approval of telisotuzumab vedotin (Emrelis) for c-Met overexpressing NSCLC expanding the addressable ADC patient population in the world's largest targeted therapy indication, Eli Lilly's March 2023 expanded Verzenio indication removing Ki-67 requirement and broadening the eligible early breast cancer patient population for CDK4/6 inhibitor targeted therapy, the accelerating FDA and EMA approval pace for biomarker-selected targeted therapies enabling faster commercial launch timelines, the progressive expansion of NGS biomarker testing into routine oncology practice globally expanding the diagnosed targetable mutation-positive patient population, and Asia-Pacific's rapidly growing cancer patient population and healthcare investment creating new regional targeted therapy demand. Valued at US$ 95.12 billion in 2024, the market is projected to reach US$ 235.68 billion by 2031, recording a CAGR of 13.84% over the 2025 to 2031 forecast period.
Request Sample Pages of this Research Study @ https://www.businessmarketinsights.com/sample/BMIPUB00031706
The market's revenue structure combines monoclonal antibody revenues from the dominant therapy type with growing ADC, TKI, and immune checkpoint inhibitor revenues, lung cancer indication revenues from the largest indication, hospital pharmacy revenues from the primary distribution channel, and North American revenues from the leading regional market supported by the highest healthcare spending and most active targeted therapy approval pipeline globally.
Cancer Targeted Therapy Market Drivers and Opportunities:
Advancements in Genomic Profiling and Biomarker Discovery Driving Cancer Targeted Therapy Market
Emergence of genomic profiling and biomarker discovery are powerful agents of growth in the cancer targeted therapy marketplace, reshaping how cancer is diagnosed, stratified, and treated. Strides are being made to move away from the one-size-fits-all approach to cancer therapy to precision medicine approaches with detailed molecular information about the molecular drivers of individual tumors. Next-generation sequencing (NGS), whole exome sequencing, and liquid biopsies have allowed scientists and clinicians to discover many actionable genetic mutations (e.g., EGFR, ALK, KRAS, BRAF, HER2) that serve as targets for variable levels of specificity and other available or developing therapies.
These discoveries have opened systems of drug development for a new class of targeted drugs (e.g., tyrosine kinase inhibitors (TKIs), monoclonal antibodies, antibody-drug conjugates (ADCs)), that seek to inhibit cancer-drivers cascades without damaging healthy tissue, in theory decrease toxicity and improve overall efficacy. Clinical relevance of biomarkers has also expanded from simply supporting treatment selection; biomarkers can often be used to provide insight to disease progression (e.g. circulating tumor DNA (ctDNA), predicting development of therapeutic resistance, or guidance on eligibility for combination therapies. Furthermore, regulatory bodies, including the FDA and EMA, have provided increasing developmental guidance and approval for biomarker-driven therapeutics often through biosimilars. From the miniature stage-one trials discussed previously, tumor-agnostic indications have been developed where drug approval is related to treatment of patient populations based on predetermined genetic alterations rather than based solely on the cancer type.
Expanding Targeted Therapies into Rare Cancers and Tumors Driven by Specific Genetic Mutations
Expanding targeted therapies into rare cancers and tumors based on specific genetic alterations is a major and growing opportunity in the cancer targeted therapy space. The development of targeted therapies has historically taken place in high-prevalence cancers, like lung, breast, and colorectal cancers, where there are large patient populations that have well-characterized mutations, and where research and clinical trial expenses are justified. Advances in genomic sequencing and molecular diagnostics have uncovered actionable mutations in a whole host of less common and even ultra-rare tumor types, and have identified new therapeutic targets that may not have been known before.
Some tumors, like cholangiocarcinoma, gastrointestinal stromal tumors (GIST), anaplastic thyroid cancer, and certain sarcomas, to name a few, have well-characterized mutations—such as FGFR2 fusions, NTRK gene fusions, RET mutations, and KIT/PDGFRA alterations—that can be directly targeted. The development of tumor-agnostic therapies, like TRK inhibitors and checkpoint inhibitors approved for use in microsatellite instability-high (MSI-H) or TMB-high tumors, exemplifies that cancers can be treated based on their genetic profile, and not whereby tissue the cancer originated from. In fact, there are regulatory pathways in place to encourage these developments, which include accelerated pathways and orphan drug designations, incentivizing pharma companies to pursue therapies for smaller patient populations.
Frequently Asked Questions
What is the Cancer Targeted Therapy Market size in 2024? The Cancer Targeted Therapy Market is valued at US$ 95.12 billion in 2024.
What is the projected Cancer Targeted Therapy Market size by 2031? The market is projected to reach US$ 235.68 billion by 2031.
What CAGR is the Cancer Targeted Therapy Market expected to register? The market is estimated to record a CAGR of approximately 13.84% from 2025 to 2031.
What factors are driving Cancer Targeted Therapy Market revenue growth? Global cancer burden creating permanent therapy demand, AbbVie's Emrelis c-Met ADC approval, Eli Lilly's Verzenio expanded indication, accelerating FDA and EMA biomarker-driven approval pace, NGS routine adoption expanding diagnosed targetable patient populations, and Asia-Pacific healthcare investment collectively drive exceptional market revenue through 2031.
About Us
Business Market Insights is a market research platform that provides subscription service for industry and company reports. Our research team has extensive professional expertise in domains such as Electronics & Semiconductor; Aerospace & Defense; Automotive & Transportation; Energy & Power; Healthcare; Manufacturing & Construction; Food & Beverages; Chemicals & Materials; and Technology, Media, & Telecommunications.
Contact Us
If you have any questions about this report or would like further information, please contact us: Contact person: Ankit Mathur Email: sales@businessmarketinsights.com Phone: +16467917070